MedTech U.S. Reimbursement Masterclass
A Strategic Roadmap for Aligning Regulatory and Reimbursement Decisions for Sustainable Payment and Adoption in the U.S. Market
Your Results: What This Means for Your Reimbursement Readiness
Mostly A's
You may feel that it’s too early to think about reimbursement, but waiting could delay market access later. Consider taking a first step to getting some early feedback on your value proposition from payers and hospital administrators to strengthen your clinical study design and increase confidence in your product.
Mostly B's
You're on the right path by considering reimbursement early. This is a valuable moment to identify and close remaining gaps by engaging stakeholders strategically to help refine your approach and support more predictable outcomes over time.
Mostly C's
You’re ahead of the curve! By thinking about reimbursement and validating your value proposition with multiple stakeholders now, you’re positioning your MedTech for coverage and faster market adoption. Continuing to make informed reimbursement decisions early helps accelerate momentum.
If your answers were mostly A's or B's, it suggests that key reimbursement decisions are still taking shape which is where many MedTech teams find themselves.
Take the Next Steps With Confidence
If reimbursement readiness matters to your U.S. strategy — and your answers suggest it does — this masterclass is designed to support your next decisions.
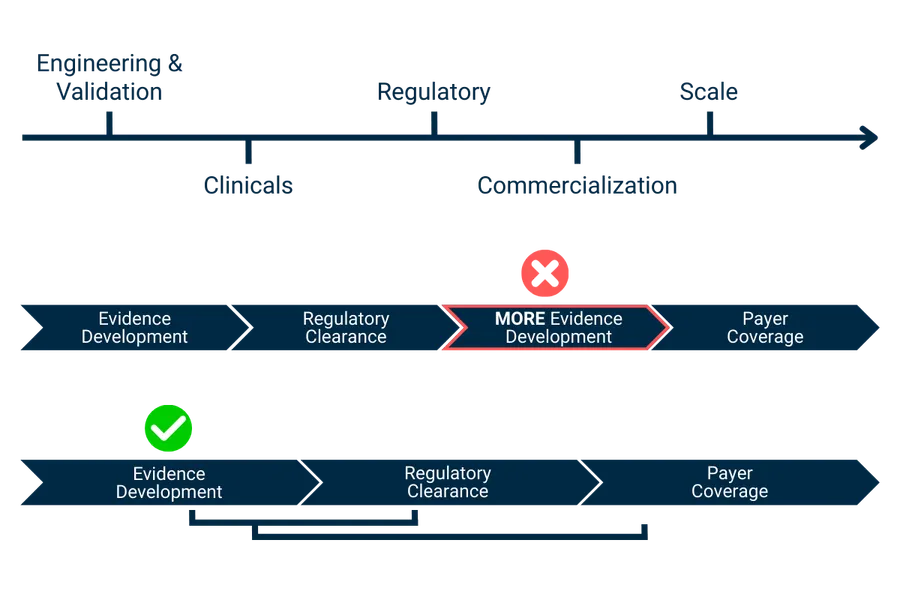
Where Your Reimbursement Strategy Fits
U.S. reimbursement isn’t determined by a single factor. It’s shaped by how clinical evidence, regulatory strategy, coding pathways, payer expectations, and stakeholder alignment come together over time. When these elements aren’t addressed early — or are considered in isolation — MedTech companies often face constraints in payment and adoption.
Need help or have questions?
Email our team at [email protected]